-
Structure and Function of Macromolecules - Organic molecules that weigh more than ___________________ daltons are referred to as macromolecules.
- These macromolecules are constructed of smaller units called _________________. These ___________________________ are subdivided into their basic units called ______________________.
-
- Making and breaking of polymers:
- Dehydration synthesis: is a _____________________ process by which two molecules are chemically bonded through the use of enzymes and a loss of _______________.
- Example: glucose + glucose = maltose +
__________.
- Hydrolysis: is a ______________________ process by which the bond between monomers are broken by the enzyme and the addition of __________.
-
Example: Sucrose + __________________ = glucose + fructose.
- The Four Major Organic Compounds found in Living Things.
-
- A. Carbohydrates: include __________________ and their polymers.
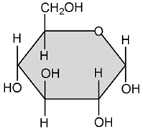
- 1. Monosaccharides: The basic formula (CH2O)
- Examples: Glucose, ____________________, and __________________ are hexose ( 6 Carbon ) sugars. _____________________ and Ribose are ( 5 carbon ) pentose sugars.
- A. Carbohydrates: include __________________ and their polymers.
-
-
-
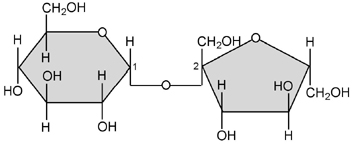
- 2. Disaccharides: These are double sugars with the formula C12H22O11. Notice that one molecule of water is missing from the formula. The covalent bond holding the two monomers together is called a glycoside linkage. Examples: sucrose = glucose + fructose; maltose = glucose + glucose; and lactose = glucose + galactose.
-
-
-
-
- 3. Polysaccharides: The basic formula is ( C6H10O5)n .
- A. Storage Polysaccharides: _______________ is a plant storage polysaccharide that is composed entirely of glucose. Amylose is the simplest form of starch. Amylopectin is more complex and is branched. Glycogen is an animal starch stored in the ________________ and muscles of vertebrates.
- B. Structural Polysaccharides: __________________ and ______________ are examples of structural polysaccharides.
- Cellulose is the most abundant organic compound on earth. It is made of glucose, like starch, but differs in the type covalent bonds.
- B. Lipids: A group of polymers that have one characteristic in common, they do not mix with __________________. They are hydrophobic. Some important groups are fats, phospholipids, and steroids.
- Fats: are large molecules composed of 2 types of monomers, __________________ ( an alcohol containing 3 carbons) and 3 ______________ ____________molecules.
- 3. Polysaccharides: The basic formula is ( C6H10O5)n .
- The two types of fatty acids are: ________________________ and _____________________________.
- The saturated fatty acids do not contain any __________________ bonds between the carbons. Unsaturated fatty acids contain one or more double bonds between their carbons. These double bonds cut down on the number of _________________ atoms that can be attached to the carbon in the molecule. This causes the molecule to bend or kink at each of the double bond sites.
-
Fatty Acids 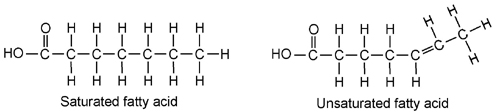
-
-
Fat -
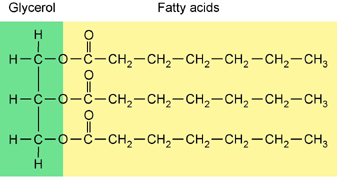
- Characteristics of Fats:
Saturated Unsaturated 1. solid at room temperature
1. liquid at room temperature
2. found mostly in animals
2. found mostly in plants
3. no double bonds between carbons
3. double bonds found between carbons
-
-
- Function of fats:
- acts as insulation in higher vertebrates,
- serves as an energy storage source 1g.=9 Kcal of energy,
- and acts as a shock absorber for internal organs.
-
- Phospholipids: structurally related to fats but contain 2 fatty acids and one molecule of _________________________. These molecules are found making up the plasma membrane of cells. They exhibit a polar and non polar quality. The phosphate group is ______________________ while the fatty acid area is _____________________________.
- Steroids: Lipids characterized by a carbon skeleton of 4 fused rings. _________________________ is an important steroid found in all animal tissue. Plants do not contain cholesterol. Cholesterol functions in many ways: it is a precursor from which many of the bodies steroids are constructed from. It also adds strength to the plasma membrane in animal cells.
- C. Proteins: macromolecules that make up _______________% of the dry weight of most cells. Their monomers are called ________________________. Most amino acids consist of a carbon bonded to an amino group, hydrogen, an R group,and a ____________________ group which makes it an acid. There are _______ different amino acids. The bond formed between amino acids is called a ___________________ bond.
- Phospholipids: structurally related to fats but contain 2 fatty acids and one molecule of _________________________. These molecules are found making up the plasma membrane of cells. They exhibit a polar and non polar quality. The phosphate group is ______________________ while the fatty acid area is _____________________________.
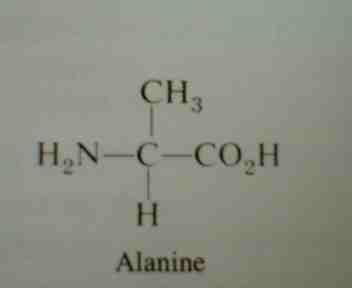
-
Types of proteins:
- 1. Structural: functions in support, examples: elastin,
collagen, and keratin
- 2. Storage: food source, examples: ovalbumin and casein
- 3. Transport: moves other substances, examples: hemoglobin and cell membrane proteins
- 4. Hormonal: coordinates bodily activities, example insulin,
- 5. Contractile: movement, examples: actin and myosin
- 6. Antibodies: defense, examples: Ig.E, IgA, and Ig.G
- 7. Enzymes: aid in chemical reactions, examples: amylase and proteases
- D. Nucleic Acids: DNA and RNA.
- _____________________ are the monomers that come together to form a nucleic acid. They contain either a ribose or deoxyribose sugar, phosphate, and a nitrogenous base ( guanine, adenine, cytosine, thymine , or uracil). Base pairing rule. A-T, A-U, C-G.
- 2. Storage: food source, examples: ovalbumin and casein
- DNA has a double helix shape, while RNA is single stranded.
-
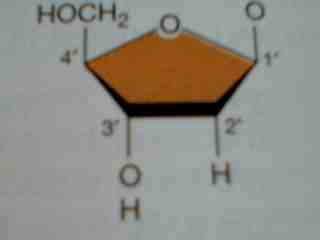
Deoxyribose Sugar
-